Contemporary nuclear armaments utilize a combination of chemical explosives, nuclear fission, and nuclear fusion. The chemical explosives compress nuclear substance, triggering fission; the fission produces a large energy output in the form of X-rays, establishing the high temperature and pressure requirements for fusion ignition.
Fission and Fusion Processes
Within each atom lies a "nucleus" (plural: "nuclei"), where neutrons and protons are tightly packed together. Most nuclei are relatively stable, signifying a consistent and unchanging composition of protons and neutrons.
During fission, the nuclei of certain larger atoms divide into smaller, lighter nuclei, liberating surplus energy. This can sometimes occur naturally, but can also be instigated externally in some nuclei. A neutron is projected at the nucleus and absorbed, resulting in instability and fission. In certain elements such as specific isotopes of uranium and plutonium, the fission process also emits additional neutrons, which can initiate a chain reaction if absorbed by nearby atoms.
Fusion operates contrarily: under extremely high temperatures and pressures, some lightweight nuclei can merge to form heavier nuclei, emitting energy in the process.
In contemporary nuclear armaments that utilize both fission and fusion, a single warhead can discharge more explosive energy within a split second than all the armaments used during the entirety of World War II, including Fat Man and Little Boy, the two atomic bombs deployed on Japan.
Understanding Their Functioning
All nuclear weapons leverage fission to cause an explosion. "Little Boy", the first nuclear weapon ever operational in warfare, functioned by propelling a hollow uranium-235 cylinder at a similar material target "plug".
Nuclear fuel
nly certain isotopes of certain elements can undergo fission (an isotope is a variation of the same element with different numbers of neutrons in the nucleus). Plutonium-239 and uranium-235 are the most common isotopes used in nuclear weapons.
Each piece by itself was not enough to constitute a critical mass (the minimum amount of nuclear material needed to maintain fission)—but by colliding the pieces, critical mass was reached and a fission chain reaction occurred.
Modern nuclear weapons work slightly differently. Critical mass depends on the density of the material: as the density increases, the critical mass decreases. Instead of colliding two sub-critical pieces of nuclear fuel, modern weapons detonate chemical explosives around a sub-critical sphere (or “pit”) of uranium-235 or plutonium-239 metal. The force from the blast is directed inward, compressing the pit and bringing its atoms closer together. Once dense enough to reach the critical mass, neutrons are injected, initiating a fission chain reaction and producing an atomic explosion.
In fusion weapons (also called “thermonuclear” or “hydrogen” weapons), the energy from an initial fission explosion is used to “fuse” hydrogen isotopes together. The energy released by the weapon creates a fireball that reaches several tens of million degrees—temperatures in the same range as the center of the sun (which also runs on fusion).
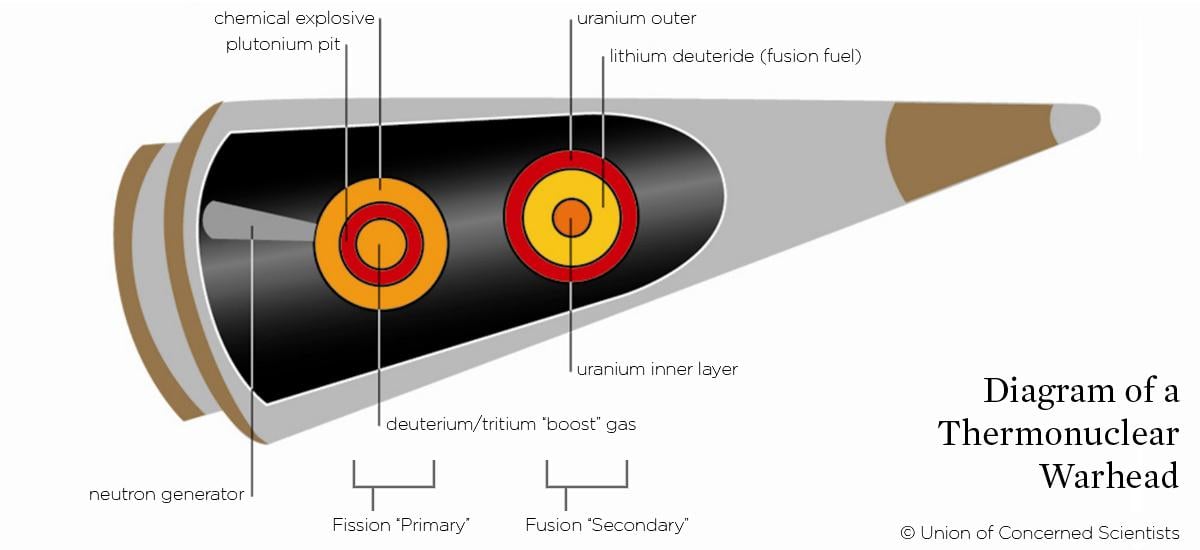
Warheads in-depth
The explosions used in thermonuclear weapons are often described as a primary (the chemical and fission explosions) and secondary (the subsequent fusion blast). However, the actual mechanisms are considerably more complicated.
For example, a pure fission primary is inefficient—the plutonium pit will blow itself apart before most of the plutonium-239 can fission. Instead, the reaction can be “boosted” by including hydrogen gas (consisting of the isotopes deuterium and tritium) in the center of a hollow pit. As the surrounding plutonium fissions, the hydrogen gas undergoes fusion and releases neutrons, inducing additional fission.
Similarly, the secondary doesn’t consist purely of fusion fuel; layered within it is a fission "spark plug," consisting of either plutonium-239 or uranium-235. As the primary explosion compresses the fuel from the outside, the spark plug material becomes supercritical and fissions, heating the hydrogen from the inside and facilitating further fusion reactions.
Fusion releases neutrons. These neutrons hit a layer of uranium surrounding the fusion fuel causing atoms in it to fission; this fissioning generally contributes more than half of the weapon’s total explosive yield.
Thermonuclear weapons that don’t include this uranium “blanket” are called neutron bombs, as the neutrons freed by fusion are released from the weapon. Neutron bombs therefore create a larger amount of radiation than a normal weapon of the same yield. During the Cold War such weapons were considered for use against tank attacks, with the goal of disabling tank crews without having to physically destroy the tank.

Natural uranium must be enriched in centrifuges like these before it's usable in nuclear weapons. Photo: Nuclear Regulatory Commission
Nuclear fuel
While a number of elements are fissionable (meaning they can undergo fission), only a few are used in nuclear weapons. Most common are the isotopes uranium-235 and plutonium-239 (reminder: isotopes are atoms of the same element that differ only in their number of neutrons).
Uranium is found throughout the world and can be mined from mineral deposits (it can also be extracted from seawater, but doing so is currently much more expensive). However, only a small fraction (less than one percent) of naturally occurring uranium is uranium-235. Producing usable uranium requires a process of “enrichment,” in which different uranium isotopes are separated and concentrated (usually using centrifuges, which operate like salad spinners). This is extremely costly, difficult, and time-consuming, and is one of the central barriers to constructing a nuclear bomb.
Plutonium can also be used, but only occurs naturally in trace amounts. It can, however, be produced as a fission byproduct in nuclear reactors, then separated *by a process called “reprocessing.” Plutonium separation is easier than uranium enrichment—it involves separating different elements, not different isotopes of the same element—but it’s a highly radioactive process that requires heavily shielded facilities with remote-handling equipment.
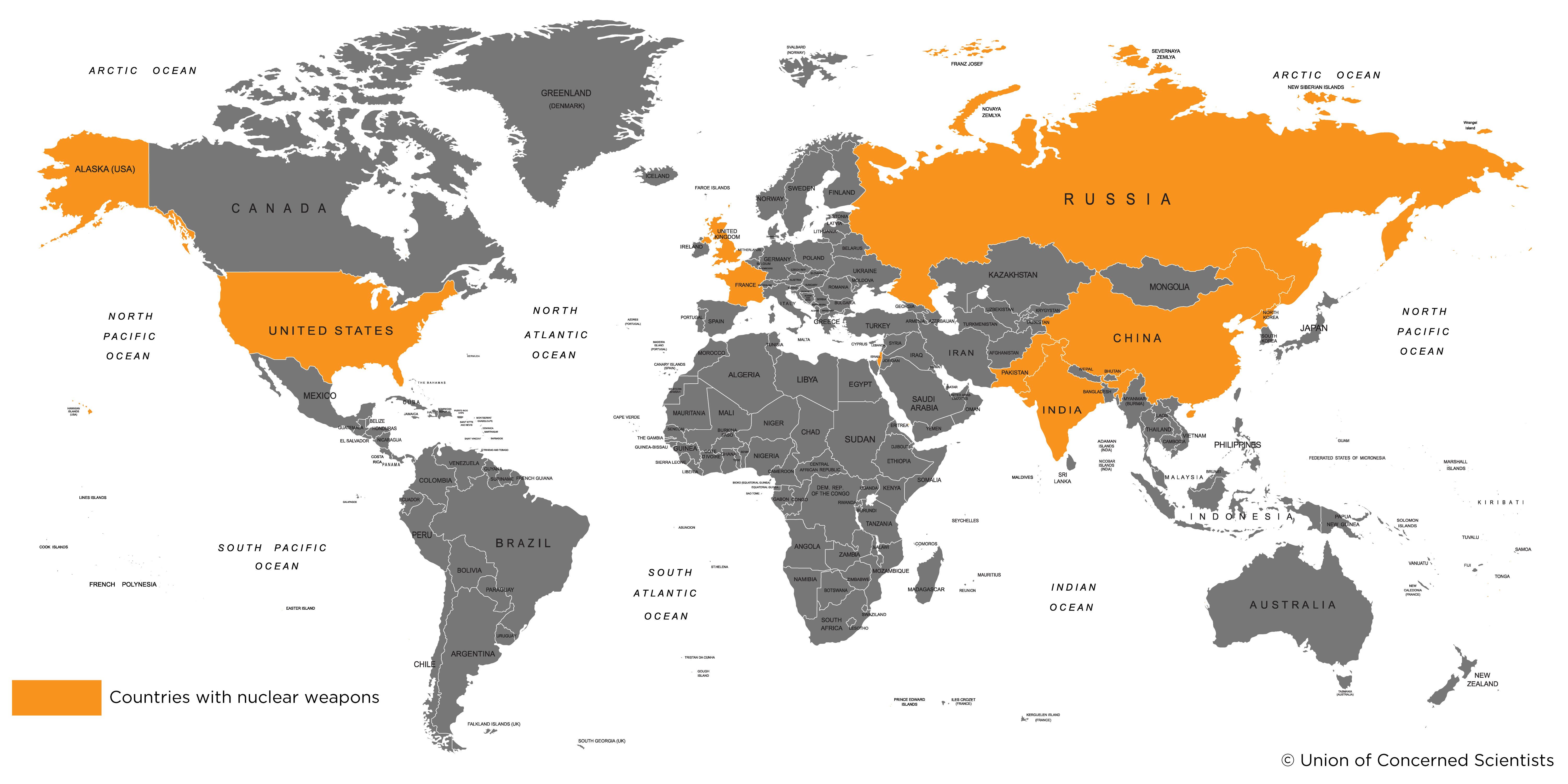
Nuclear weapons are currently held by nine nations: the United States, Russia, the United Kingdom, France, China, India, Pakistan, Israel, and North Korea. The United States pioneered the development of these formidable weapons, detonating the first atomic bomb in 1945 and subsequently the first hydrogen bomb in "Operation Ivy" in 1952. As of current estimates, the U.S. has around 5,428 nuclear warheads. The Soviet Union, now Russia, followed suit in 1949 and currently possesses around 5,997 nuclear warheads, the highest of any nation. France, China, and the UK also possess nuclear weapons, with estimated stockpiles of around 300, 260, and 215 warheads respectively [2]. India and Pakistan, with approximately 120 and 130 warheads respectively, are also part of this exclusive club. Israel, while not officially acknowledging its nuclear capabilities, is estimated to have around 80 warheads. The exact extent of North Korea's nuclear capabilities remains uncertain, with estimates suggesting they possess between 5 to 10 weapons.


Post a Comment